Description
- It is intended to aid in the rapid diagnosis of SARS-CoV-2 infections.
- The Healgen Covid-19 Antigen Test (Swab) does not differentiate between SARS-CoV and SARS-CoV-2.
- Each kit contains everything required to carry out the tests, no additional equipment is needed.
- Approved by Public Health England & validated by the UK government.
- Results in 15 minutes.
- Each kit includes clear instructions and is easy to use.
- Cannot be shipped to a USA address.
The Healgen Covid-19 Antigen Test is one of only three UK Government approved Rapid Antigen Test. It is in high demand from schools, councils and companies.
- This test is being rolled out by the UK government to mass test the population in Liverpool and other cities, click here for UK Government approval (test 3 on the list).
- This test is designed for use by clinical professionals (eg Doctor/Practice Nurse/Employer Occupational Health depts etc).
- An application for exceptional use of a COVID-19 rapid test, to be used by members of the public for home use, was approved on the 23rd December 2020 by the Medicines and Healthcare products Regulatory Agency (MHRA)
Clinical Evaluation:
- Positive Percent Agreement (PPA): 97.3%
- Negative Percent Agreement (NPA): 99.6%
- Overall Percent Agreement (OPA): 98.9%
- Specimen: Nasopharyngeal (NP) Swab
- Time to Results: 15 minutes
- Shelf Life: usually 24 months from the date of manufacture
Benefits:
- Rapid testing for SARS-CoV-2 antigen within 15 minutes
- Facilitates patient treatment decisions quickly
- Simple, time-saving procedure
- All necessary reagents provided & no equipment needed
- High sensitivity and specificity
Each kit contains:
- 20 Test cassettes
- 20 Sterile swabs
- 20 Extraction tubes and dropper tip
- 1 Workstation
- 2 Buffers
- 1 Package insert
Performance Characteristics:
- The Healgen Covid-19 Antigen Test (Swab) has been evaluated with specimens obtained from patients.
- A commercialised molecular assay was used as the reference method.
- The results show that the Healgen Covid-19 Antigen Test (Swab) has a high overall relative accuracy.
Test Procedure:
1. Specimen Collection
- Use the nasopharyngeal swab supplied in the kit
- Carefully insert the swab into the nostril of the patient, reaching the surface of posterior nasopharynx that presents the most secretion under visual inspection.
- Swab over the surface of the posterior nasopharynx.
- Rotate the swab several times. Withdraw the swab from the nasal cavity.

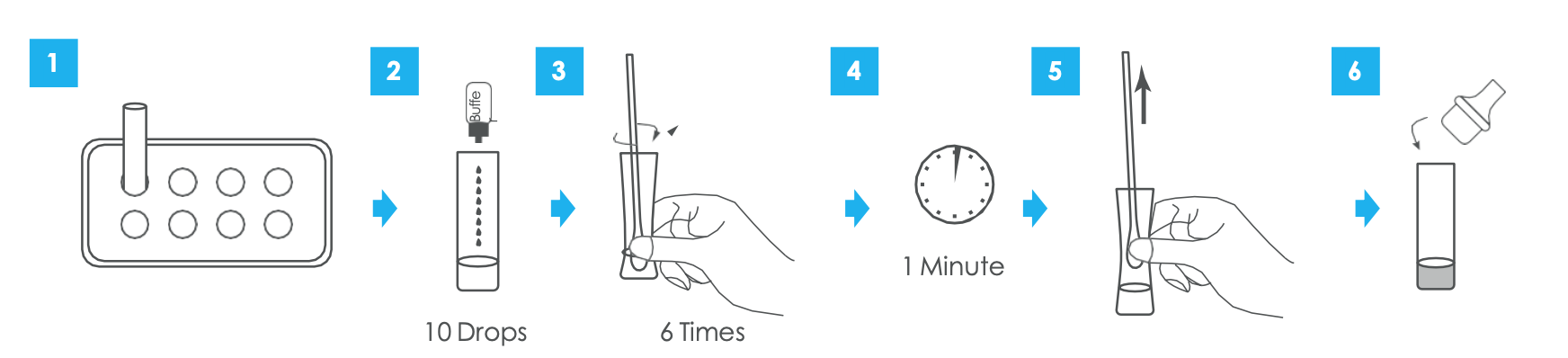
2. Sample preparation
- Insert the test extraction tube into the workstation
- Make sure that the tube is standing firm and reaches the bottom of the workstation
- Add 0.3 mL (about 10 drops) of the sample extraction buffer into the extraction tube.
- Insert the swab into the extraction tube which contains 0.3 mL of the extraction buffer. Roll the swab at least 6 times while pressing the head against the bottom and side of the extraction tube. Leave the swab in the extraction tube for 1 minute.
- Squeeze the tube several times with fingers from outside of the to immerse the swab
- Fit the dropper tip with filter on top the extraction tube tightly
- Remove the swab. The extracted solution will be used as test sample.
3. Interpretation of Results
- Allow the test device, test sample and buffer to equilibrate to room temperature prior to testing
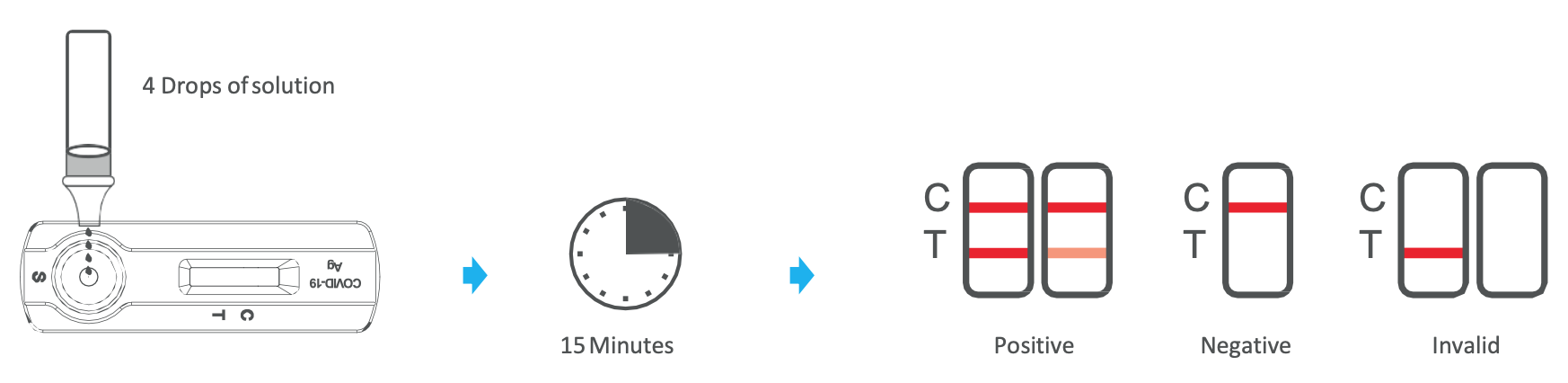
- Remove test device from the sealed pouch just prior to the testing and lay flat on work bench.
- Ensure the nozzle with filter is fitted on to the sample extraction tube tightly.
- Reverse the sample extraction tube, and add 4 drops (about 100 μL) of test sample by squeezing the extracted
- solution tube into the sample window.
- Wait for the colored band(s) to appear. The result should be read in 15 minutes.
- Do not interpret the result after 20 minutes.
Test results:
- POSITIVE – The presence of two lines as control line (C) and test line (T) within the result window indicates a positive result.
- NEGATIVE* – The presence of only control line (C) within the result window indicates a negative result.
- INVALID – If the control line (C) is not visible within the result window after performing the test, the result is considered invalid. Some causes of invalid results are because of not following the directions correctly or the test may have deteriorated beyond the expiration date. It is recommended that the specimen be re-tested using a new test.
Note: The intensity of color in the test line region (T) may vary depending on the concentration of analytes present in the specimen. Therefore, any shade of color in the test line region (T) should be considered positive. Please note that this is a qualitative test only and cannot determine the concentration of analytes in the specimen. Insufficient specimen volume, incorrect operating procedure or expired tests are the most likely reasons for control band failure.
(*Negative results do not rule out SARS-CoV-2 infection, particularly those who have been in contact with the virus. Follow up testing with a molecular diagnostic test should be considered to rule out infection in these individuals.)
In-vitro use only. Please note, each box includes 1 x work station and 2x buffer solutions, therefore it can not be split up.
Returns Policy: This product is non-returnable and non-refundable.
- All persons intending to use the Deliverables must rely on their own knowledge and judgment in the selection and use of those Deliverables; and
- Antibodies and related products have a limited usable life and require storage and use in controlled conditions.
The Buyer:
- is responsible for complying with any applicable legislation and regulations governing the test;
- shall use and handle the product in accordance with applicable laws, including applicable health and safety regulations; and
- shall ensure that it and/or its personnel handling the tests have read and understood the instructions to ensure accuracy of such tests.
- is responsible for complying with any potential requirements set out by national and local authorities which may have to be adhered to as a result of undertaking the tests (ie. any reporting requirements and isolation requirement).
Technical Details about this test
The Healgen Covid-19 Antigen Test (Swab) is an in vitro immunochromatographic assay for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2 in nasopharyngeal (NP) swab specimens directly or after the swabs have been added to viral transport media from individuals who are suspected of COVID-19 by their healthcare provider. It is intended to aid in the rapid diagnosis of SARS-CoV-2 infections. The Healgen Covid-19 Antigen Test (Swab) does not differentiate between SARS-CoV and SARS-CoV-2.
Storage: The kit can be stored at room temperature or refrigerated (2-30°C), please follow instructions on each pack. The test device is stable through the expiration date printed on the sealed pouch. The test device must remain in the sealed pouch until use. Please read instructions fully before starting any of the tests. DO NOT FREEZE. Do not use beyond the expiration date.
Reviews
There are no reviews yet.